 
This absorbance value can then be used to determine the concentration of a given element (or atoms) within the sample. The difference between the intensity of the light emitted from the source (e.g., lamp) and the light collected by the detector yields an absorbance value. If the wavelength of the source light has energy corresponding to the energy difference between two energy levels in the atoms, a portion of the light will be absorbed. In atomic absorption spectroscopy, light of a predetermined wavelength is passed through a collection of atoms. The type of atoms present in a sample, or the amount of atoms present in a sample can be deduced from measuring these changes in light wavelength and light intensity.Ītomic spectroscopy is further divided into atomic absorption spectroscopy and atomic emission spectroscopy. Ca, Na, etc.) and thus will absorb/emit photons in a correspondingly unique pattern. 
Because each element has a unique number of electrons, an atom will absorb/release energy in a pattern unique to its elemental identity (e.g. In optical spectroscopy, energy absorbed to move an electron to a higher energy level (higher orbital) and/or the energy emitted as the electron moves to a lower energy level is absorbed or emitted in the form of photons (light particles). Electrons may move between orbitals, but in doing so they must absorb or emit energy equal to the energy difference between their atom's specific quantized orbital energy levels. Atomic orbitals are quantized, meaning they exist as defined values instead of being continuous (see: atomic orbitals). Atomic absorption spectrometers are one of the most commonly sold and used analytical devices.Įlectrons exist in energy levels (i.e. Because optical spectroscopy is often less expensive and has performance adequate for many tasks, it is far more common. This complexity translates into higher purchase costs, higher operational costs, more operator training, and a greater number of components that can potentially fail. Mass spectrometry generally gives significantly better analytical performance, but is also significantly more complex. In the latter case, the main division is between optical and mass spectrometry. It can be divided by atomization source or by the type of spectroscopy used. Since unique elements have unique emission spectra, atomic spectroscopy is applied for determination of elemental compositions. In physics, atomic spectroscopy is the study of the electromagnetic radiation absorbed and emitted by atoms. 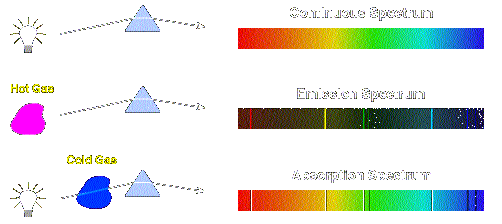
JSTOR ( September 2008) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed.įind sources: "Atomic spectroscopy" – news Please help improve this article by adding citations to reliable sources. This article needs additional citations for verification. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
